 Other sources of gamma radiation include particle and anti-particle annihilations, and nuclear fusion.įusion reactions release more energy than any other nuclear reaction, requiring huge amount of energy input for the reaction to occur in the first place. The mass deficit for excited nuclei are generally not provided. The energy associated through the emission of a gamma photon from an excited nucleus is comparatively quite small. 
As with alpha decay, there is the potential for a gamma photon to be emitted from the nucleus if it is left in an excited state. In agreement with the conservation of momentum, the beta particle is released at a very large velocity relative to the barely impacted decayed nucleus, due to the enormous difference in size. The energy produced in beta decay is very similar to alpha decay, although at a smaller scale. The larger nucleus is sometimes left in an excited state, emitting a gamma photon to enter a more stable configuration. To satisfy the conservation of momentum, this results in the significantly smaller alpha particle being emitted with a much greater magnitude of velocity than the decayed nucleus. The energy released in this reaction generally increases the kinetic energy of the products. Large, unstable nucleus usually undergo alpha decay, involving the emission of an alpha particle (He-4 nucleus) from the parent nucleus. This will always be provided in the question, the values in the periodic table cannot be used. When undertaking quantitative analysis of a nuclear reaction it is important to remember that the atomic mass must be used. As such, this means that has mass “lost” (termed the mass deficit) is energy that has been converted into other forms, namely heat (kinetic) and light. 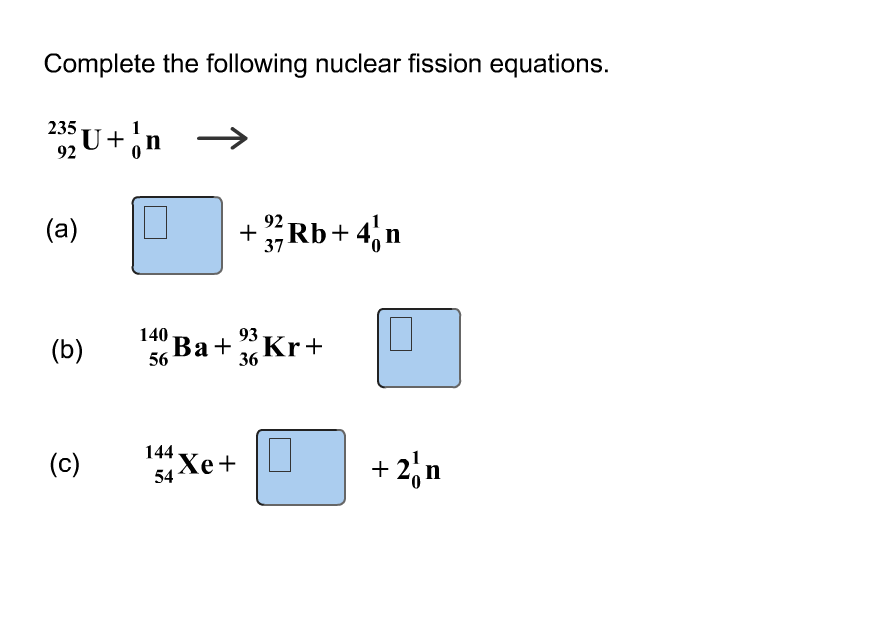
Considering the relationship between mass and energy, described by E=mc 2, this would mean that the conservation of energy has been violated, which is not possible. Through nuclear reactions, there is an apparent loss of mass between the reactants and products. Quantitative Analysis of Nuclear Reactions – LearnĪll quantitative analysis of nuclear reactions relies on two key principles, the Law of Conversation of Energy and Einstein’s mass-energy equivalence. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
